The Hill Top Chamber® is a patented occlusive patch test system widely used to evaluate the safety and skin compatibility of products that come into contact with human skin. This standardized device allows controlled application of test materials to the skin, ensuring reliable assessment of irritation, sensitization, and overall dermatological safety. The Hill Top Chamber system is commonly used in studies involving cosmetics, fragrances, pharmaceuticals, soaps, detergents, fabrics, films, and other consumer products to support product safety and regulatory requirements.
System Design / Options
The basic component of the Hill Top Chamber System is a molded plastic chamber flexible enough to conform to the contour of the skin. This chamber has a unique double rim, consisting of an outer ring and an inner flexible flange, for a close fit to the skin surface to increase occlusivity. Within the chamber a non-woven Webril® pad holds the test sample. The chamber is sandwiched between squares of release paper and semi-occlusive, hypoallergenic adhesive tape, ready for use. The Hill Top Chamber can be provided in single-unit form or in multi-unit strips with any number of chambers. Virtually any size or shape of chamber can be custom produced.
Plastic chambers alone, with or without Webril® pad are also available.
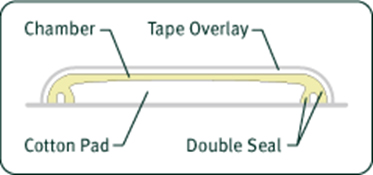
Chamber Composition
The Hill Top Chamber is manufactured using Kraton® G-2705, a product of the Shell Oil Company. Kraton® meets or exceeds the current USP requirements for physico-chemical and biological tests for Class VI polymers. The chamber is soft and flexible thus providing cohesion between the test material and the skin sites. The Kraton® material has been approved for use by the FDA. Other plastics may be provided based on client need. Hill Top Chamber Systems are backed with Durapore®, a "silk-like" tape with a latex-free, hypoallergenic adhesive manufactured by 3M that offers high strength and adhesion. Webril® inserts are 100% cotton.
19 mm
Plain
Plain W/Pad
Full System
25 mm
Plain
Plain W/Pad
Full System
Chamber Sizes / Configurations
| Standard/Custom | Chamber Diameter | Tape Size | Chamber Capacity | Variations Currently Available* |
|---|---|---|---|---|
| Standard | 19 mm 25 mm |
3x3 cm 4.5 x 4.5 cm |
.2 ml .3 ml |
Plain, Plain w/Pad, Full System |
| Custom | 33 mm 52.34 mm |
5.5x5.5 cm 7.5 x 5.5 cm |
2.0 ml 2.5 ml |
Domed Plain |
* Virtually any size or shape of chamber can be custom produced.
